Written by MaryBetts Sinclair, author of Hydrotherapy for Bodyworkers: Improving Outcomes with Water Therapies.
In combination with skilled touch, water treatments are perhaps the very oldest and most revered of all healing modalities. Pain from injuries, issues from damaged muscles and joints, circulation problems, chronic tension, chronic pain and emotional stress have long inspired healers to relieve suffering this way.
From ancient Rome’s great baths to Russian saunas, Indian Ayurvedic steams, Native American sweat lodges, Turkish baths and Japanese hot springs, peoples the world over use and love hydrotherapy and massage together. In Germany, the warm waters of Baden-Baden have been used for over eight thousand years, and in Bath, England, for ten thousand. 2800 years ago, Irish sweat houses made of sod and stone were used for rheumatism.
As a bodywork student or practitioner, you may be wondering what is the advantage of adding water treatments to your skill set. Here’s how it can improve your effectiveness:
*** Like massage, hydrotherapy can relieve discomfort and pain, stimulate the flow of blood and lymph, and make connective tissues more pliable and comfortable to the touch.
***Hydrotherapy is soothing and stress-reducing. The ancients realized the effect depression and stress can have upon a person, and over centuries, chronic depression was called everything from gloom or melancholia to neurasthenia or dysthymia. In ancient Greece, while warriors bathed to reduce fatigue and promote wound healing, warm baths were also ordered to relieve “dejection and low spirits.” The founder of modern psychiatry, Philippe Pinel, (1745-1826) recommended warm baths to calm “overwrought nerves.”
In the 1800’s, many fashionably wealthy Victorians who suffered from depression spent their lives travelling to spas in hope of a cure, especially those diagnosed with mania, hysteria and other “nervous disorders”. For the first half of the twentieth century, hydrotherapy was a mainstream treatment for mental institutions, complete with bathtubs, steam baths and fomentation stations. Treatments were intended to make mentally ill patients more comfortable and compliant, and thus less likely to be violent. Fomentations were soothing, while hours-long baths calmed restless or agitated patients. But in the late 1940’s, even though the American Medical Association still affirmed the value of spa therapy for “nervous conditions,” the introduction of psychotropic medications put an end to the use of hydrotherapy and massage for sufferers.
So what is it about hydrotherapy that helps release stress and tension?
According to cognitive psychologist John Bargh, physical feelings of warmth are linked early in life to feelings of safety, and we subconsciously associate physical warmth with emotional warmth. “Especially with animals that breastfeed their infants, the experience of being fed and held and protected goes hand in glove with feelings of warmth and closeness…the positive response to heat is hardwired into our brains.” This helps to explain the deep enjoyment and relaxation our clients experience when we use any warm treatment before or during a session.
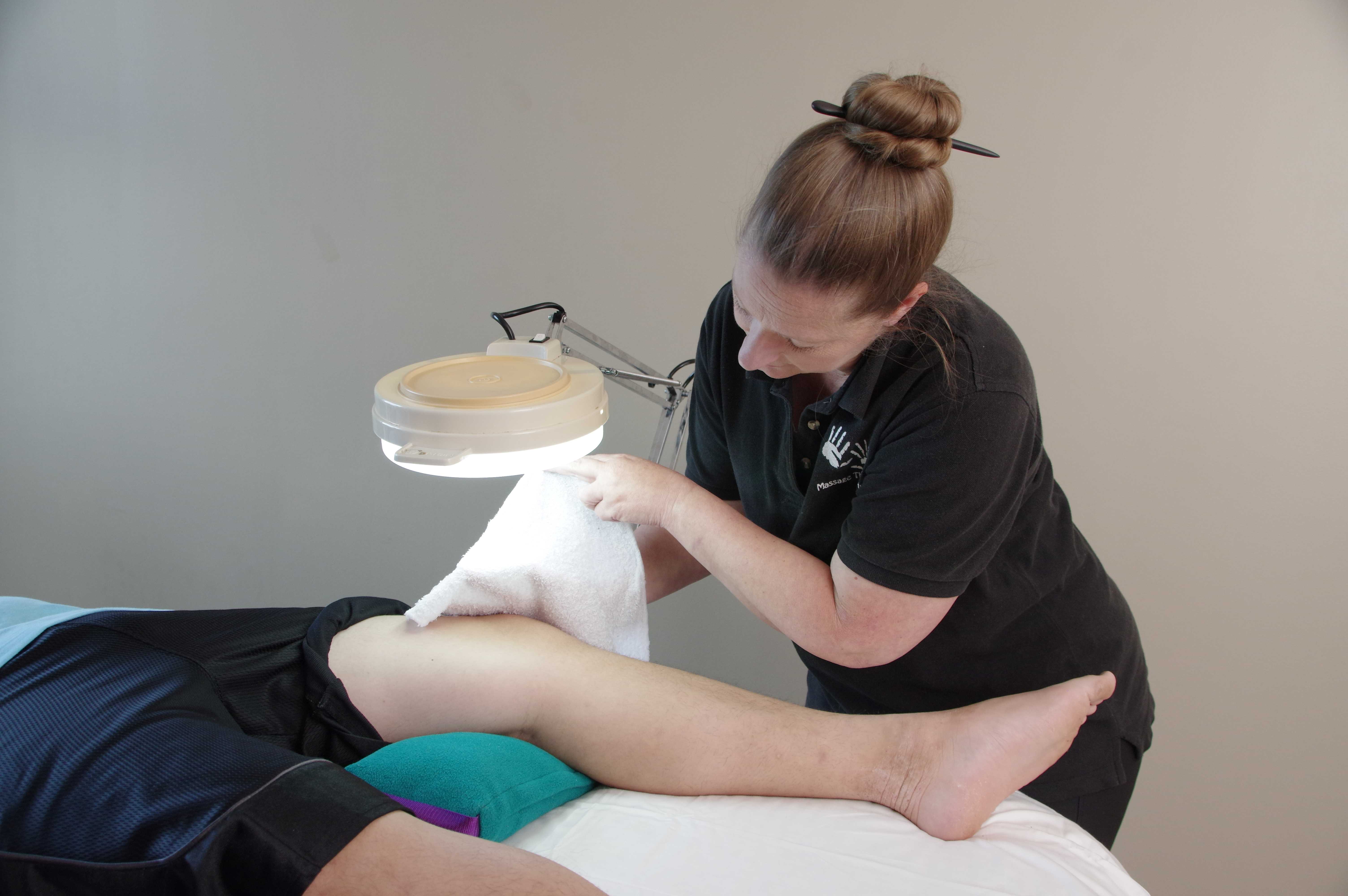
Whole-body heating has antidepressant effects because it activates specific brain areas that are important for the regulation of mood and body temperature and so a steam bath, sauna, or even a warm shower before a session will help the client calm and settle before the bodywork begins. A simple warming body wrap can be made with blankets and the client’s head, neck and feet massaged while the wrap is in place. Partial-body treatments have that soothing effect as well. For example, a warm moist pack over the spine, a paraffin dip for arthritic hands, warm compresses over the face for TMD, a heat lamp or heating pad over a painful knee, and many other local heat treatments can help clients feel safe and relaxed and get the most out of their sessions. When clients arrive for sessions chilled, tense or uptight, a warm treatment is a great help and also a treat for them.
Because hydrotherapy is so beloved, new treatments are being invented all the time, such as flotation therapy for chronic pain, Watsu for relaxation, water exercise baths for tiny hospitalized premies, and special hyperthermia treatments for depression. With such a popular modality, likely more new water treatments will be brought forward as time goes on.
For a deeper dive into hydrotherapy and water therapies, click here to learn more about Hydrotherapy for Bodyworkers by MaryBetts Sinclair.
To stay in the loop about all things Singing Dragon and Handspring, sign up to our mailing list here: https://pages.hachette.co.uk/sguk-newsletter-sign-up/.